Chemistry #2 – Elements, Atoms and Chemical Changes
Define and explain physical changes
Physical changes are changes that happen to a certain object in a physical manner, such as ice turning into water, and chemical change is when some chemical turns into another chemical, such as iron rusting.
Changes of State
Physical changes are changes that happen to a certain object in a physical manner, such as ice turning into water, and chemical change is when some chemical turns into another chemical, such as iron rusting.
Changes of State
Define each of the six changes of state:
Freezing: liquid to solid
Melting: solid to liquid
Evaporation: liquid to gas
Condensation: gas to liquid
Deposition: solid to gas
Sublimation: gas to solid
Freezing: liquid to solid
Melting: solid to liquid
Evaporation: liquid to gas
Condensation: gas to liquid
Deposition: solid to gas
Sublimation: gas to solid
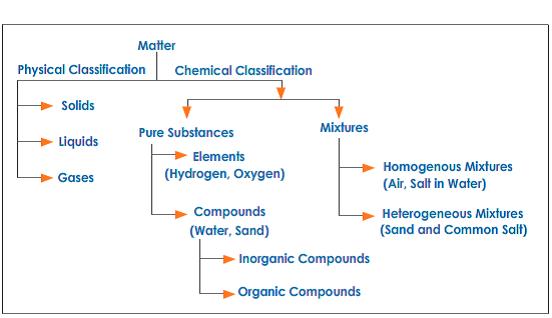
What are pure substances? (Give three examples)
Carbon Dioxide, Hydrogen, Iron
What are elements and compounds? (Give three examples with pictures for each)
Elements are a type of pure substance, and is all shown on the periodic table. Compounds are pure substances with more than one type of chemical in it. Such includes:
Uranium Titanium Plutonium
Carbon Dioxide, Hydrogen, Iron
What are elements and compounds? (Give three examples with pictures for each)
Elements are a type of pure substance, and is all shown on the periodic table. Compounds are pure substances with more than one type of chemical in it. Such includes:
Uranium Titanium Plutonium
What are mixtures?
A mixture is made from two or more substances, but are not combined chemically.
How many types are there?
Homogenous Mixtures and Heterogeneous Mixture
Give three examples with pictures
Water Sand The air we breath
A mixture is made from two or more substances, but are not combined chemically.
How many types are there?
Homogenous Mixtures and Heterogeneous Mixture
Give three examples with pictures
Water Sand The air we breath
How are chemical changes different from physical changes?
Chemical changes are when two 0r more substances or chemicals bond, and creates a new chemical (burning a piece of paper). Physical change is when a substance or chemical changes in a physical way, but not chemically (ice turning into water).
Give five examples of a chemical change with pictures
Metal rusting
Milk going sour
Paper burning
Glow stick lighting up
Ice pack activating
Chemical changes are when two 0r more substances or chemicals bond, and creates a new chemical (burning a piece of paper). Physical change is when a substance or chemical changes in a physical way, but not chemically (ice turning into water).
Give five examples of a chemical change with pictures
Metal rusting
Milk going sour
Paper burning
Glow stick lighting up
Ice pack activating
What are the five clues that a chemical change has occurred?
Color Change
Gas Is Given Off
Change in temperature
Solid Forms (Precipitate)
Light Is Given Off
Color Change
Gas Is Given Off
Change in temperature
Solid Forms (Precipitate)
Light Is Given Off
Baking Soda and Vinegar:
The baking soda is the base and the vinegar is the acid. When the two meets they form C02 bubbles and creates water. The reaction: a gas is released and a new chemical is formed. This is also impossible to reverse.
Milk and Dish Soap:
Food colouring is dropped onto the surface of the water to make the reaction more obvious. The soap does not sink, and when it spreads on the surface, it grabs onto the food coloring. This is impossible to reverse.
Elephant's Toothpaste:
The reaction is endothermic because it produces heat. It is a foamy substance caused by the rapid decomposition of hydrogen peroxide. This is impossible to reverse.
The baking soda is the base and the vinegar is the acid. When the two meets they form C02 bubbles and creates water. The reaction: a gas is released and a new chemical is formed. This is also impossible to reverse.
Milk and Dish Soap:
Food colouring is dropped onto the surface of the water to make the reaction more obvious. The soap does not sink, and when it spreads on the surface, it grabs onto the food coloring. This is impossible to reverse.
Elephant's Toothpaste:
The reaction is endothermic because it produces heat. It is a foamy substance caused by the rapid decomposition of hydrogen peroxide. This is impossible to reverse.